
The post-pandemic world is realizing that the intersection of technology and a traditional way of doing business already exists with us. The highly competitive nature of our business only exemplifies the need of incorporating innovative technologies to augment and support customers in the ever-evolving Regulatory landscape. Trends show that the global automation market, especially in the life sciences industry, is poised to grow close to $2.5 bn by 2025 with a CAGR of approximately 7.5%. Many companies are already moving in this direction and gaining focus by incorporating investments to strengthen and protect their strategic assets like Regulatory information.
Regulatory affairs divisions at life sciences companies must deal with a lot of manual processes. Monitoring dossiers for drugs and/or devices across different Health Authorities in various stages of their “Lifecycle,” updating files for manufacturing or labeling, etc., are arduous tasks with little or no scope of oversight or misses. Non-compliance and sub-optimized performance are simply unacceptable since they have an adverse implication on not just the financial aspect, but also the brand reputation. Employees should be able to easily access documents and information while compiling reports for Regulatory purposes. Automation tools driven by Artificial Intelligence (AI)/Machine Learning (ML)/Robotic Process Automation (RPA) do not just provide quick and reliable data collection, but also faster access and better compilation of data when it is most needed. Digital transformation and implementation of new technologies to counter the existing and the upcoming challenges is the only way forward.
Digital transformation trends in the biotech and pharma Regulatory market point towards an infusion of innovation and automation tools available, thereby transforming traditional service delivery leading to exponential and immediate value-addition to the customers. They consist of simple, creative, and quickly deployable innovative toolsets that lead towards a transformational process or task improvement with significant benefits and outcomes that can be realized immediately.
With AI, ML, NLP, and other automation tools, companies can improve efficiency, enhance productivity, ensure safe drugs and devices, hasten market entry, and remain in those markets, ultimately driving better business outcomes.
Some of the processes in Regulatory functions where incremental and disruptive innovation are proving to be increasingly helpful are:
- CTD Auto File Mapping
- Global Classification and Registration Widget for Medical Devices
- Publishing Automation
- Smart Translation of Scientific Documents
- Content to Carton Platform with Automated Label/Artwork Content Generation
- Individual Case Study Report (ICSR) Automation
- Clinical Performance Report (CPR) Automation for Medical Devices
- NLP-based Quality Control of Medical Writing Function
- Automated Translation Bots for Pre-defined Clinical Documents
In addition to these intelligent product suites that the strategic partners offer customers; they also assist clients in moving the automation scale forward. Consulting services for the evaluation of processes and automating routine operational activities and scientific activities from traditional processes across safety, medical writing, labeling, clinical & Regulatory streams are the top trends that will transform the biotech and pharma Regulatory market.
Incremental Innovation Category | Cross Functions | Automation Tool Description |
Smart Content and Document Processing | Artwork | Auto annotations of Artwork PDFs from given inputs |
Clinical Safety, Regulatory Quality, and Compliance, MA | QC/formatting standards for any word documents and NLP-based QC using Amazon Comprehend | |
Regulatory Operations | Automated document-level publishing and submission-level eCTD publishing of dossiers | |
Smart Search and Translation Automation | Safety, Medical Writing, Labeling, Clinical, and Regulatory | Amazon web-services-based translation/transcoding of scientific documents |
Bulk multilingual keyword and contextual search | ||
Robotic Process Automation | Medical Device Regulatory Affairs | Global Medical Device classification tracker |
Safety, Medical Writing, Labeling, Clinical, and Regulatory | Smart web crawlers for real-time screening for new journals and articles | |
Global Regulatory Intelligence | RPA/NLP based internal and external decision support systems |
The applicability of automation in some other functional areas can be best described as below:
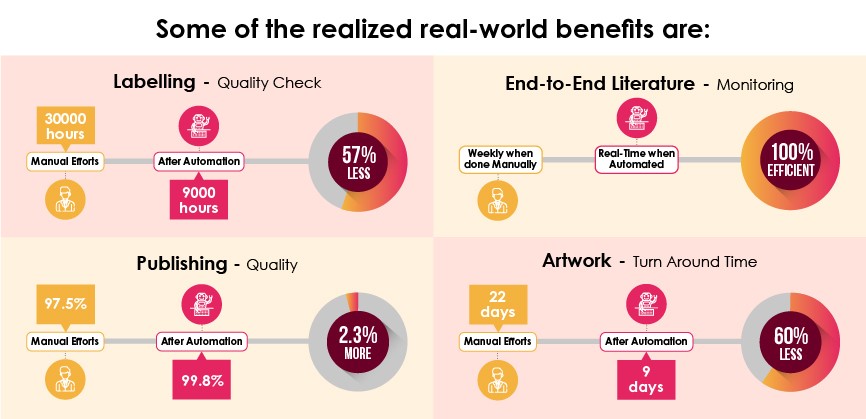
To guide companies through the entire process of digital transformation from creating a strategy, to discovering redundant processes, designing optimal automation, testing, and full-scale use as well as effectiveness monitoring, it is best to partner with a proven Regulatory consulting partner.
Experts in Regulatory automation can assist pharmaceutical, life sciences, consumer products, and medical device companies with their digital transformation efforts by:
- establishing and developing an automation strategy
- evaluating, comprehending, and documenting redundant business processes
- establishing operational models breaking the silos
- determining technical infrastructure needs for deploying state-of-the-art automation solutions
- training employees and assisting in the creation of an “automation-friendly” environment, in which each team member seeks out additional automation opportunities to aid in the company’s digital transition. By educating team members (train-the-trainer) on how to create, operate, and manage automation solutions, the companies become self-sufficient in a short time
- using digital software, attain quick resolution to challenges that arise in day-to-day Regulatory compliance activities
- round the clock support for the company's adoption of automation technology
- assisting in the planning and establishment of Regulatory Automation Center of Excellence or Digital Transformation Centers to facilitate organization-wide automation processes
If you are not sure where to begin or how to improve your company's process automation and digitization with cutting-edge technological solutions, consult a strategic Regulatory partner like Freyr Digital on how to approach your company’s AI/ML/RPA automation effort.
