
A multitude of challenges are faced by the Regulatory departments by the move from paper-based to eCTD submissions that continues around the world. Most of the life science companies are struggling hard to keep up with the USFDA standard in using eCTD format and meet the specified deadline. The compulsion of the eCTD format in the US and Canada has enhanced the submission procedure and made it easier by bridging the gap between the time and market which in turn is helping in minimizing expenses on electronic submissions to the pharmaceutical industries. Though the benefits offered in this kind of submission are many, it is still quite challenging for the life science companies. Some of the setbacks are discussed below which are faced in the USFDA eCTD submission publishing:
- Annual Reports - The eCTD doesn’t easily allow the annual report to be authored as one document for this approach and can lead to validation errors and issues and can set the application for a rejection.
- Clinical Study Reports (CSRs) -The preparation of a CSR allows the reviewers to easily navigate and track so that even if the section is updated the report need not be replaced.
- Hyperlinking - One of the most common issues that publishers face due to typographical errors during document authoring is the attempt to hyperlink a section which might not exist at all.
- Report Numbering - The main issue when a study data scan takes place across multiple reports it is important to insert a unique study ID to ensure that if there are any cross-references in summary sections referring to the content, they are set clear and easily identified.
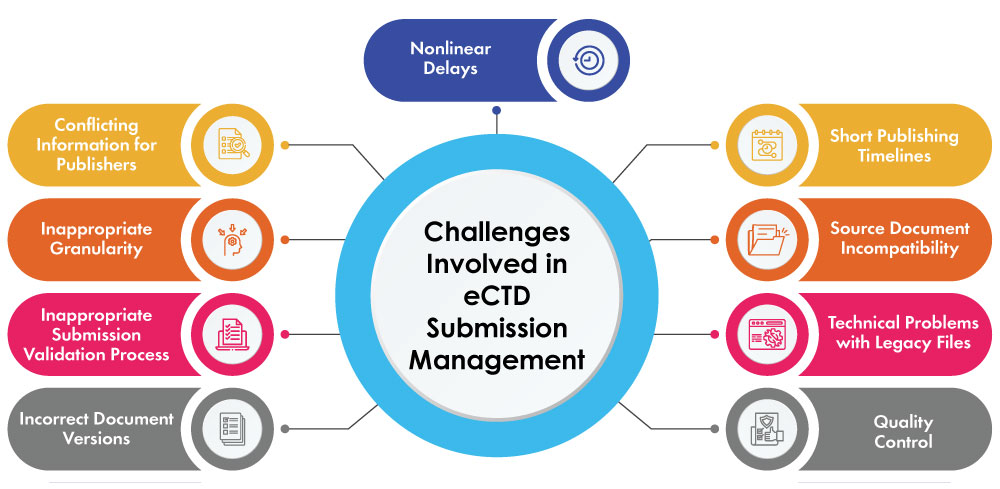
Challenges Involved in eCTD Submission Management
Pharmaceutical companies are under immense pressure dealing with the logistical challenges of managing IT applications and systems to submit error-free documents within stringent timelines. This becomes an even bigger challenge when the costs increase or cause delays to the submission’s approval, and at worst result in receipt of a refusal to file. Even though the publishers might be highly experienced, Regulatory publishing teams typically encounter the following problems (Click here for your reference):
- Conflicting Information for Publishers: Depending upon the experience of the Regulatory team, critical information required in the publishing process is necessary to showcase to the publishers but might also be ambiguous even though it is included in the content of the submission. For example, eCTD submissions rely heavily on the use of metadata which are included in critical capacities such as folder paths in the final eCTD which provides additional information about the elements. An easy way to prevent potential rework is by using well-designed procedures and forms, by providing this information to publishers at the same time as the source files.
- Inappropriate Granularity: An eCTD publishing document produced using a quality template with the appropriate level of granularity has a huge effect on publishing. On planning to submit a section as multiple leaves, they should be supplied as the corresponding number of source documents rather than being complied into a single file for segregating at the time of publishing. Every source document that must be sent back for reformatting is another small opportunity for the project to be delayed.
- Inappropriate Submission Validation Process: An inappropriate validation may arise if there are any system or publisher errors while downloading the files. The main benefit of the eCTD is the ability to check its technical conformity upon submission by the applicant or the agency.
- Incorrect Document Versions: To complete publishing of a submission with the right document or document version with more intensive QC reviews is the goal. Stringent procedures are required for the groups that use file shares for publishing repositories. Publishing teams that use a closed Document Management System (DMS) in their publishing workflows generally avoid this issue as only those versions marked as approved can be published.
- Nonlinear Delays: Not only are delays sometimes inevitable but they also can result in adverse effects on the submission timelines. Given a situation, the slot for publishing the project cannot be moved back by even a day or two due to delay of other projects, then it is bound to extend the delay even further.
- Short Publishing Timelines: Due to an exceedingly long process of submission, time lost in previous stages of the process often is expected to be recovered during publishing. One of the most time-saving technique of leveraging the risk in the submission process is by publishing independently the modules or sections of the submission.
- Source Document Incompatibility: During the process of scanning source documents to automatically extract information differing from Word/PDF file types and tools from different vendors, source files are scanned and elements such as internal document links, existing bookmarks, and heading/outline styles are processed and collected into the software’s database to create bookmarks and hyperlinks in the published output. If source files are not set up as per the publishing software expectancy, this process might take extra time may be required post-publishing to manually add navigational elements.
- Technical Problems with Legacy Files: Although in the most critical times the legacy files may have been printed without issue in the past, electronic publishing is extremely efficient in highlighting technical issues. The issues are generally not difficult to solve but are time-consuming and cause a delay knowing the fact that the most important tool in the publisher’s toolbox is time. Such a situation can be addressed well before hand by publishing submissions teams using incremental builds to catch up on time that are causing issues in the process.
- Quality Control: By the time publishing begins at the right time, source file content should be final and approved, as changing a document during the publishing process can extend the project timeline even further. A few of the quality control points that needs to be set clear throughout the project to ensure these points are appropriate to the task to make an eCTD submission are:
- All source documents should be mandatorily quality checked before entering the publishing workflow
- The submission structure (the assembly/outline) within the publishing software should be independently reviewed prior to any publishing
- All the published PDF files must be reviewed on the screen
- Check bookmarks and links in published PDF files
- Always validate the eCTD submissions prior to any submission
Freyr Digital has a solution to all the challenges listed above for your organization’s publishing and submission-level activities. Freyr SUBMIT PRO, a cloud-hosted eCTD software, makes it easy for our Regulatory operations team and eCTD publishing specialists to provide additional support when you need it the most. Freyr ensures efficient, timely submissions through our eCTD software supported by an in-built eCTD viewer and validator.
Would you like to learn more? Request a demo.
